This implies that the work performed on the system is converted into heat that is supplied by the system to the surroundings. The observation is in accordance with the first law: if the state of the system does not change (the temperature is constant), the internal energy of the system is constant and, from equation (8), it is found that the amount of heat which flows from the system to the surroundings is equal to the amount of work performed on the system. This can only be accomplished by a flow of heat from the system to the surroundings. Suppose work is performed on the paddle wheel, but the system is kept at constant temperature. In section 3.3, it was demonstrated that a system, consisting of an amount of water in which a paddle wheel is placed, may be heated by performing work on the paddle wheel (i.e. The experience that leads to the second law is the following. 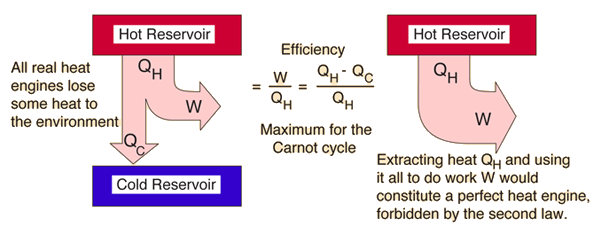
For didactic purposes, however, the first law is usually treated first. 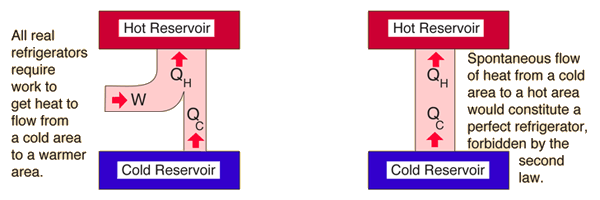
The second law of thermodynamics was developed during the investigations of Sadi Carnot (1796 - 1832) and dates from before the first law. van Ekeren, in Handbook of Thermal Analysis and Calorimetry, 1998 4.1 Negative formulation of the second law
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
